A new study from scientists at La Jolla Institute for Immunology (LJI) gives researchers a guide to neutralizing Lassa virus using a trio of rare antibodies isolated from survivors of Lassa virus infection.
Lassa virus is a deadly virus endemic to West Africa, where it is primarily spread by rodents. The virus causes Lassa fever, a disease that strikes up to 300,000 each year and typically starts with flu-like symptoms, but can lead to severe illness, death, and long-lasting symptoms, such as deafness. For pregnant women Lassa virus is especially dangerous: nearly 90% of infections during pregnancy are fatal.
LJI scientists can now show exactly how a cocktail of three human antibodies can block viral infection. These antibodies may prove valuable in upcoming clinical trials for Lassa therapies, and the LJI team plans to use their new map of the Lassa virus surface glycoprotein to design a much-needed vaccine.
“We now know where these three therapeutic antibodies act and how exactly they act,” says Kathryn Hastie, Ph.D., an LJI Instructor and the Director of the Antibody Discovery Center at LJI.
The findings were published in Science Translational Medicineas a cover story on October 26, 2022. The research was led by the Saphire Lab at LJI, including Instructor Haoyang Li, Ph.D., Hastie and Professor Erica Ollmann Saphire, Ph.D., in collaboration with Luis Branco, Ph.D., of Zalgen Labs LLC.
The power of neutralizing antibodies
In 2017, Hastie and her colleagues in the Saphire Lab (then at Scripps Research) published the first-ever structural images of the Lassa virus glycoprotein. Lassa uses glycoproteins to enter host cells and initiate infection. Hastie’s glycoprotein structure gave researchers an idea of what they were up against.
Hastie’s breakthrough came as researchers hunted for the rare human antibodies that could break through Lassa’s defenses. The hope was for researchers to harness these neutralizing antibodies to develop Lassa fever therapeutics or vaccines.
This hope became reality when research partners at Tulane University and Zalgen Labs LLC isolated a promising group of Lassa-fighting antibodies from the blood of Lassa fever survivors. Collaborators from the University of Texas Medical Branch went on to test a cocktail of three neutralizing antibodies in non-human primates. This antibody therapy, called Arevirumab-3, proved 100 percent effective in treating Lassa fever, even in animals with advanced disease.
“This was a groundbreaking finding,” says Saphire. “The dogma had been that antibodies would not be protective against Lassa virus.”
When it came time to test the cocktail in human clinical trials, the researchers faced a problem. The U.S. Food and Drug Administration was not prepared to launch clinical trials until the researchers could uncover the mechanism that made the therapy so effective. Exactly how did these neutralizing antibodies target Lassa virus glycoprotein and prevent infection?
To answer this question, researchers needed a more detailed map of Lassa glycoprotein. Hastie’s original structure of the glycoprotein required complicated molecular engineering to provide sufficient stability for imaging. Her structure gave scientists a critical glimpse of Lassa glycoprotein, but not the full picture. Plus, some of the promising therapeutic antibodies could not recognize this or any version of engineered Lassa glycoprotein. Researchers needed to isolate a natural glycoprotein target for further investigation.
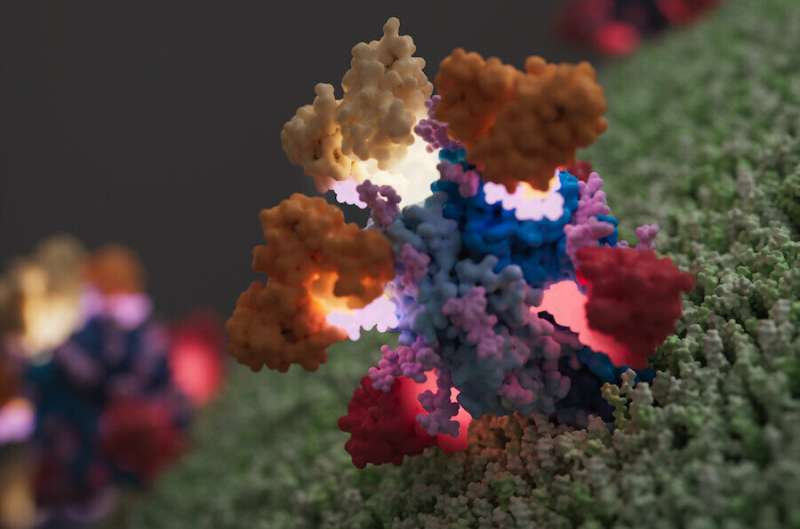
Fortunately, the Saphire Lab had the tools and expertise to reveal these molecular details. Li led the effort to produce a “native” Lassa glycoprotein. Thanks to advances in protein production and three years of perseverance, Li’s version of the glycoprotein was a copy of the real thing and could be recognized by all three antibodies used in Arevirumab-3. Li then used a technique called cryo-electron microscopy single-particle analysis to image the native glycoprotein together with the three antibodies.
“Haoyang’s ingenuity and hard work enabled us to see the structures we couldn’t see before,” says Hastie.
A new map of Lassa virus targets
Based on the high-resolution structures and several functional assays, the team revealed exactly how the three antibodies used in Arevirumab-3 neutralize Lassa virus.
Meet neutralizing antibodies: 8.9F, 12.1F, and 37.2D.
Hastie was amazed to see how antibody 8.9F binds to the very top of the glycoprotein structure. This area of the glycoprotein is where three molecules (called protomers) come together to form a “trimer,” a kind of twisted trefoil, as Hastie describes it. Lassa would normally use this region of the glycoprotein to bind with receptors on host cells, but Li’s structure shows how a single 8.9F jumps in and binds to all three protomers simultaneously to block infection.
“The structure is really a beautiful illumination of how this antibody essentially mimics the host receptor to block the glycoprotein receptor from binding,” says Hastie. “It’s an absolutely gorgeous structure to see.”
Meanwhile, the neutralizing antibody called 12.1F binds to just one protomer in the three-sided trimer. Fortunately, any therapeutic would have many copies of 12.1F. Moving as a team of three, each 12.1F antibody can bind to a protomer to aid in neutralizing the virus.
At the same time, copies of antibody 37.2D target Lassa virus by binding in a way that anchors adjacent protomers together. This antibody activity is a huge problem for Lassa, since the virus needs to open up its trimer (where the protomers come together) to infect host cells. With 37.2D on the scene, its entry machinery is locked up, unable to function.
“Lassa has another trick. It shields itself using a thick coat of human carbohydrate molecules—like a wolf in sheep’s clothing,” says Saphire, “Haoyang’s structures clearly show how these potent, protective antibodies breach or even utilize the carbohydrates to target and neutralize the virus.”
“The findings fill a critical gap in Lassa virus research and may pave the way for Arevirumab-3 to move into clinical trials.” says Branco, who will lead the Zalgen team to perform the future clinical trials.
Navigating Lassa’s weak spots
With this new study, the researchers have a guide to better targeting three Lassa weak spots (called epitopes). Two of these critical epitopes had never been mapped before.
In fact, just this year, the Saphire Lab has published three articles on anti-Lassa neutralizing antibodies (including this paper). The other two investigations were published in Cell Reports and mBio.
“This body of work now offers the first-ever full-epitope map, revealing every vulnerable target of the Lassa glycoprotein,” says Saphire.
“We now have a very clear idea about the neutralizing epitope surface and the requirements needed on the glycoprotein for anybody binding and recognition,” adds Hastie.
Source: Read Full Article
